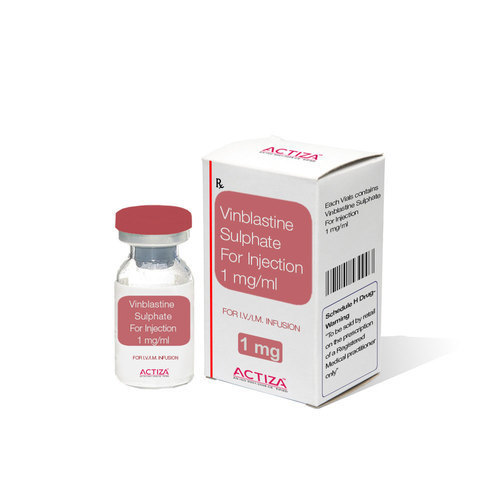
Vinblastine Sulphate Injection
Product Details:
- Indication Anticancer, used in the treatment of Hodgkins disease, lymphomas, and certain other cancers
- Salt Composition Vinblastine Sulphate
- Dosage Form Injection
- Enzyme Types Other
- Feature Other
- Ingredients Vinblastine Sulphate, Water for Injection
- Application Other
- Click to View more
Vinblastine Sulphate Injection Price And Quantity
- 100 Box
- 1 USD ($)/Box
Vinblastine Sulphate Injection Product Specifications
- Other
- Clear, colorless solution
- Vinblastine Sulphate
- Other
- Injection
- Vinblastine Sulphate, Water for Injection
- 2 Years
- Other
- Store at cool and dry place.
- Anticancer, used in the treatment of Hodgkins disease, lymphomas, and certain other cancers
- Glass vial with rubber stopper and aluminum seal
- Prescription Only
- Administer under supervision of a qualified physician experienced in cancer chemotherapy
- Intravenous (IV)
- As per Indian Pharmacopeia/IP standards
- Hypersensitivity to Vinblastine or related compounds
- 10 mg / 10 ml
- Vial
Vinblastine Sulphate Injection Trade Information
- surat
- Delivery Point (DP), Cash on Delivery (COD), Paypal, Letter of Credit (L/C), Western Union, Letter of Credit at Sight (Sight L/C), Cash Against Delivery (CAD), Telegraphic Transfer (T/T), Days after Acceptance (DA), Cash in Advance (CID), Cheque, Cash Advance (CA)
- 1000000 Box Per Month
- 4 Days
- Contact us for information regarding our sample policy
- 1 mg/ml Bottle
- Australia, North America, South America, Eastern Europe, Western Europe, Middle East, Africa, Asia, Central America
- Jammu and Kashmir, Chandigarh, Himachal Pradesh, Andaman and Nicobar Islands, Pondicherry, Daman and Diu, Dadra and Nagar Haveli, Lakshadweep, South India, East India, Assam, Arunachal Pradesh, Bihar, Goa, Jharkhand, Karnataka, Madhya Pradesh, Maharashtra, Mizoram, Meghalaya, Manipur, Punjab, Rajasthan, Sikkim, Nagaland, Telangana, Tripura, Uttarakhand, West Bengal, Delhi, Gujarat, Haryana, Tamil Nadu, North India, Andhra Pradesh, Kerala, Central India, Odisha, West India, Chhattisgarh, Uttar Pradesh, All India
- ISO/GMP/c-GMP/WHOGMP/USFDA/EUGMP.
Product Description
Since last manyA years SaintroyA LifescienceA is leading manufacture, export andsupplier ofA Vinblastine Sulphate InjectionA from Surat, Gujarat, India. Vinblastine Sulphate Injection used with other medications, to treat a number of types of cancer.
Tags: Vinblastine Sulphate Injection manufacturer, Vinblastine SulphateA Injection exporter, Vinblastine Sulphate Injection supplier, and Vinblastine SulphateA Injection, whogmp approved Vinblastine Sulphate Injection manufacturer andA supplier, Vinblastine Sulphate Injection distributor, Vinblastine Sulphate InjectionA shipper, Vinblastine Sulphate Injection 1 mg Vinblastine Sulphate InjectionA A 1 mgA supplier, Vinblastine Sulphate InjectionA 1 mg.
ISO/GMP/cGMP/WHOGMP/NAFDAC approved, PPB Kenya approved/USFDA approved/EUGMP approved/MOH Iran approved manufacturing facility inA Gujarat, India.
Registration: Saintroy LifescienceA is interested inregistration of own brand or customer brands in ministryA of health of clientsA country and we also provide the products samples, COA, COPP, ACTD or CTD dossier, DMF, Manufacturing license, Free sales certificate and other required documents.
Export CountryA :Saintroy LifescienceA Vinblastine Sulphate InjectionA export to USA, Canada, Germany, Europe, France, Ghana, Nigeria,Kenya, Senegal, Sudan,UAE, Kuwait, Dubai, Cameroon, Lebanon, Peru, Chile,Somalia, Philippines, Myanmar, Maldives, Thailand, Singapore, Malaysia, HongKong, China, Zimbabwe, Togo, Burkina Faso, Niger,Guinea Bissau, Congo,Democratic of republic, Ivory Costa, Oman, Guatemala, Nicaragua, Salvador,Qatar, Costa Rica, Panama, Cuba, Colombia, Bolivia, Argentine, Algeria, Guyana,Morocco, Jordan, Bahrain, Iran, Egypt, Kyrgyzstan, Uzbekistan, Kazakhstan.A
Precise IV Delivery for Cancer Treatment
Vinblastine Sulphate Injection is designed for precise intravenous use in the management of various cancers, including Hodgkin's disease and lymphomas. Each vial delivers a measured dose under controlled conditions, helping oncologists tailor therapy to individual patient needs. The clear, colorless solution ensures clarity and purity, prepared as per Indian Pharmacopeia requirements.
Safe Handling and Quality Assurance
Manufactured and packaged with attention to pharmaceutical standards, the injection is secured in glass vials, rubber stoppers, and aluminum seals to ensure integrity. Its regulatory certification and prescribed storage ensure long-term efficacy, while rigorous quality control during production guarantees safety and compliance for wholesalers, dealers, and healthcare providers across India.
FAQ's of Vinblastine Sulphate Injection:
Q: How should Vinblastine Sulphate Injection be administered?
A: Vinblastine Sulphate Injection must be given intravenously by a qualified medical professional experienced in cancer chemotherapy to ensure correct dosage and minimize risks.Q: What types of cancers is Vinblastine Sulphate Injection used for?
A: This injection is primarily indicated for the treatment of Hodgkin's disease, lymphomas, and certain other cancers as prescribed by your oncologist.Q: When should Vinblastine Sulphate Injection not be used?
A: This medication is contraindicated in individuals with known hypersensitivity to Vinblastine or related vinca alkaloids. Always inform your physician about any allergies before receiving this treatment.Q: Where should I store Vinblastine Sulphate Injection?
A: Store the injection in a cool, dry place as directed, keeping it away from direct sunlight and out of reach of children. Follow specific storage recommendations provided by your supplier or healthcare provider.Q: What precautions should be followed during administration?
A: Vinblastine Sulphate Injection must be handled and administered under the supervision of a medical specialist trained in administering chemotherapy agents. Appropriate monitoring is essential throughout the process.Q: What is the benefit of using a prescription-only Vinblastine Sulphate Injection?
A: Being prescription-only ensures that Vinblastine Sulphate Injection is used safely under professional supervision, maximizing therapeutic benefits while minimizing potential side effects or misuse.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
Other Products in 'Anticancer Injection' category
 |
SAINTROY LIFESCIENCE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |