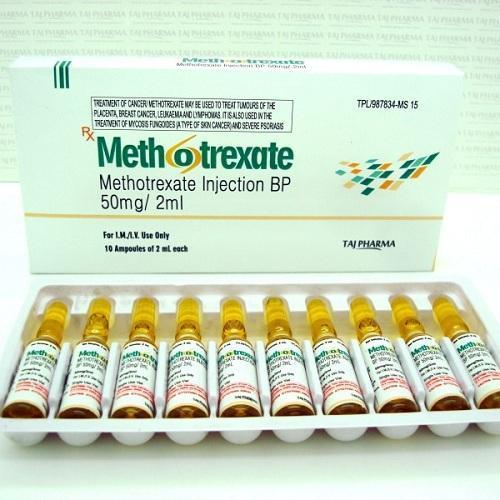
Methotrexate Injection
Product Details:
- Indication Treatment of cancer including leukemia, lymphoma, breast cancer, and certain autoimmune diseases such as rheumatoid arthritis and psoriasis
- Dosage Form Injection
- Salt Composition Methotrexate
- Enzyme Types Other
- Feature Other
- Ingredients Methotrexate, sterile water for injection, excipients
- Application Other
- Click to View more
Methotrexate Injection Price And Quantity
- 100 Box
- 1 USD ($)/Box
Methotrexate Injection Product Specifications
- Treatment of cancer including leukemia, lymphoma, breast cancer, and certain autoimmune diseases such as rheumatoid arthritis and psoriasis
- Methotrexate, sterile water for injection, excipients
- 2 Years
- Other
- Clear yellow solution
- Injection
- Methotrexate
- Store at cool and dry place.
- 7.0 - 9.0
- Other
- Other
- Odorless
- Available in 50 mg/2 mL, 500 mg/20 mL vials
- Contraindicated in pregnancy, breastfeeding, severe renal or hepatic impairment
- Monitor liver, renal function, and blood counts regularly
- Intravenous, intramuscular, or intrathecal
- Prescription only medicine
- If required, reconstitute with provided solvent under aseptic conditions
- Compatible with recommended diluents only
- Dispose according to local regulations for cytotoxic agents
- Glass vial with flip-off seal
Methotrexate Injection Trade Information
- surat
- Cash Against Delivery (CAD), Cash on Delivery (COD), Letter of Credit (L/C), Western Union, Paypal, Delivery Point (DP), Letter of Credit at Sight (Sight L/C), Telegraphic Transfer (T/T), Days after Acceptance (DA), Cash in Advance (CID), Cheque, Cash Advance (CA)
- 1000000 Box Per Month
- 4 Days
- Contact us for information regarding our sample policy
- 10 Amp / Box
- Western Europe, Australia, North America, Eastern Europe, Middle East, Africa, Central America, South America, Asia
- Assam, Dadra and Nagar Haveli, Chandigarh, Himachal Pradesh, Sikkim, Manipur, Meghalaya, Uttarakhand, Daman and Diu, Lakshadweep, South India, East India, West India, Andaman and Nicobar Islands, Arunachal Pradesh, Bihar, Delhi, Gujarat, Goa, Haryana, Jammu and Kashmir, Jharkhand, Karnataka, Kerala, Madhya Pradesh, Maharashtra, Mizoram, Nagaland, Odisha, Punjab, Pondicherry, Rajasthan, Tamil Nadu, Telangana, Tripura, Uttar Pradesh, West Bengal, All India, North India, Andhra Pradesh, Central India, Chhattisgarh
- ISO/GMP/c-GMP/WHOGMP/USFDA/EUGMP.
Product Description
Since last manyyears Saintroy Lifesciencesleading manufacture, export and supplierofMethotrexate Injectionfrom Surat, Gujarat, India. Methotrexate Injection used to treat cancer, autoimmune diseases, and ectopic pregnancy and for medical abortions.
Tags: Methotrexate Injection manufacturer, Methotrexate Injection exporter,Methotrexate Injection supplier, and Methotrexate Injection, whogmp approved Methotrexate Injectionmanufacturer and supplier, Methotrexate Injection distributor, Methotrexate Injectionshipper, Methotrexate Injection 2 ml Methotrexate Injection 2 ml supplier,Methotrexate Injection 2 ml.
ISO/GMP/cGMP/WHOGMP/NAFDAC approved, PPB Kenya approved/USFDA approved/EUGMP approved/MOH Iran approved manufacturing facility inGujarat, India.
Registration: Saintroy Lifescience is interested inregistration of own brand or customer brands in ministry of health of clientscountry and we also provide the products samples, COA, COPP, ACTD or CTD dossier, DMF, Manufacturing license, Free sales certificate and other required documents.
Export Country :Saintroy Lifescience Methotrexate Injection Exportto USA, Canada, Germany, Europe, France, Ghana, Nigeria, Kenya, Senegal,Sudan,UAE, Kuwait, Dubai, Cameroon, Lebanon, Peru, Chile, Somalia, Philippines,Myanmar, Maldives, Thailand, Singapore, Malaysia, Hong Kong, China, Zimbabwe,Togo, Burkina Faso, Niger,Guinea Bissau, Congo, Democratic of republic, IvoryCosta, Oman, Guatemala, Nicaragua, Salvador, Qatar, Costa Rica, Panama, Cuba,Colombia, Bolivia, Argentine, Algeria, Guyana, Morocco, Jordan, Bahrain, Iran,Egypt, Kyrgyzstan, Uzbekistan, Kazakhstan.
Versatile Treatment Options
Methotrexate Injection is recognized for its effectiveness across diverse medical fields, particularly oncology and autoimmune therapy. Its applications range from various cancers such as leukemia and breast cancer, to challenging autoimmune conditions including rheumatoid arthritis and psoriasis. Healthcare professionals value its flexible administration options and well-established efficacy.
Handling, Preparation & Storage
This medication must be handled by healthcare professionals under strict aseptic conditions. Reconstitution, if needed, involves the use of provided solvent and adherence to safety protocols for cytotoxic drugs. The vials are designed with flip-off seals for secure handling, and the product should be stored in a cool, dry place. Unused or expired vials require disposal according to local cytotoxic waste regulations.
FAQ's of Methotrexate Injection:
Q: How should Methotrexate Injection be administered?
A: Methotrexate Injection can be administered intravenously, intramuscularly, or intrathecally, depending on the patient's diagnosis and doctor's recommendation. Administration must only be performed by qualified healthcare professionals in a controlled medical setting.Q: What conditions is Methotrexate Injection commonly prescribed for?
A: This medication is prescribed for the treatment of various cancers such as leukemia, lymphoma, and breast cancer. It is also indicated for certain autoimmune diseases including rheumatoid arthritis and severe psoriasis.Q: When should Methotrexate Injection not be used?
A: Methotrexate Injection is contraindicated during pregnancy and breastfeeding, and in patients with severe renal or hepatic impairment. Always inform your healthcare provider of your medical history before starting this therapy.Q: Where can Methotrexate Injection be obtained?
A: Methotrexate Injection can only be obtained with a valid prescription from licensed medical practitioners. It is supplied by authorized dealers, distributors, and healthcare facilities across India.Q: What is the process for preparing Methotrexate Injection for use?
A: If reconstitution is required, it must be done with the provided sterile solvent under aseptic conditions, as per the manufacturer's instructions. Healthcare professionals should ensure compatibility with recommended diluents only.Q: How should Methotrexate Injection vials be stored and disposed of?
A: Store Methotrexate Injection vials in a cool, dry place away from direct sunlight. Disposal must adhere to local regulations for cytotoxic agents to ensure environmental and personal safety.Q: What precautions are necessary during Methotrexate therapy?
A: Regular monitoring of liver and renal function, as well as blood counts, is essential during treatment. Patients should attend all scheduled check-ups and report any unusual symptoms immediately.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
Other Products in 'Anticancer Injection' category
 |
SAINTROY LIFESCIENCE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |