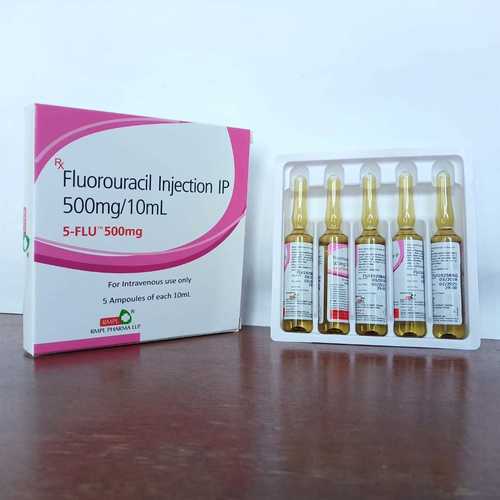
Fluorouracil Injection
Product Details:
- Indication Antineoplastic chemotherapy, used to treat cancers such as colon, rectal, breast, stomach, and pancreatic cancer
- Salt Composition Fluorouracil
- Dosage Form Injection
- Enzyme Types Other
- Feature Other
- Ingredients Other
- Application Other
- Click to View more
Fluorouracil Injection Price And Quantity
- 100 Box
- 1 USD ($)/Box
Fluorouracil Injection Product Specifications
- Other
- Clear, colorless to faint yellow solution
- Other
- Other
- Injection
- Fluorouracil
- Odorless
- Store at cool and dry place.
- Antineoplastic chemotherapy, used to treat cancers such as colon, rectal, breast, stomach, and pancreatic cancer
- 2 Years
- Other
- 8.6 to 9.0
- Meets United States Pharmacopeia standards
- 500 mg/10 mL (50 mg/mL)
- WHO-GMP, ISO certified facilities
- Available in 10 mL & 20 mL vials
- Glass ampoule or vial
- Use under medical supervision only
- Prescription only
- Do not mix with acidic solutions
- Intravenous
- Compatible with 5% dextrose or 0.9% sodium chloride
Fluorouracil Injection Trade Information
- surat
- Cash Against Delivery (CAD), Cash on Delivery (COD), Cash Advance (CA), Letter of Credit (L/C), Telegraphic Transfer (T/T), Paypal, Western Union, Letter of Credit at Sight (Sight L/C), Delivery Point (DP), Days after Acceptance (DA), Cash in Advance (CID), Cheque
- 1000000 Box Per Month
- 4 Days
- Contact us for information regarding our sample policy
- 5 Ampules Of 10 ml
- Australia, North America, Western Europe, Middle East, Central America, South America, Asia, Africa
- Himachal Pradesh, Uttarakhand, Daman and Diu, Dadra and Nagar Haveli, Lakshadweep, South India, East India, West India, Andaman and Nicobar Islands, Assam, Arunachal Pradesh, Bihar, Chandigarh, Goa, Haryana, Jammu and Kashmir, Jharkhand, Karnataka, Madhya Pradesh, Maharashtra, Mizoram, Meghalaya, Manipur, Punjab, Pondicherry, Rajasthan, Sikkim, Tamil Nadu, Telangana, Tripura, West Bengal, Nagaland, Delhi, Gujarat, North India, Andhra Pradesh, Kerala, Central India, Odisha, Chhattisgarh, Uttar Pradesh, All India
- ISO/GMP/c-GMP/WHOGMP/USFDA/EUGMP.
Product Description
Since last manyA years SaintroyA LifescienceA is leading manufacture, export andsupplier ofA Fluorouracil InjectionA from Surat, Gujarat, India. Fluorouracil Injection used for treatment of colorectal cancer, oesophageal cancer, stomach cancer, pancreatic cancer, breast cancer, and cervical cancer.
Tags: Fluorouracil Injection manufacturer, Fluorouracil Injection exporter,Fluorouracil Injection supplier, and Fluorouracil Injection, whogmp approved Fluorouracil InjectionA manufacturer and supplier, Fluorouracil Injection distributor, Fluorouracil InjectionA shipper, Fluorouracil Injection 10 ml Fluorouracil Injection A A 10 mlA supplier,Fluorouracil Injection 10 ml.
ISO/GMP/cGMP/WHOGMP/NAFDAC approved, PPB Kenya approved/USFDA approved/EUGMP approved/MOH Iran approved manufacturingA facilityin Gujarat, India.
Registration: Saintroy LifescienceA is interested inregistration of own brand or customer brands in ministryA of health ofclients country and we also provide the products samples, COA, COPP, ACTD or CTDossier, DMF, Manufacturing license, Free sales certificate and other requireddocuments.
Export CountryA :Saintroy LifescienceA FluorouracilInjection export to USA, Canada, Germany, Europe, France, Ghana, Nigeria,Kenya, Senegal, Sudan,UAE, Kuwait, Dubai, Cameroon, Lebanon, Peru, Chile,Somalia, Philippines, Myanmar, Maldives, Thailand, Singapore, Malaysia, HongKong, China, Zimbabwe, Togo, Burkina Faso, Niger,Guinea Bissau, Congo,Democratic of republic, Ivory Costa, Oman, Guatemala, Nicaragua, Salvador,Qatar, Costa Rica, Panama, Cuba, Colombia, Bolivia, Argentine, Algeria, Guyana,Morocco, Jordan, Bahrain, Iran, Egypt, Kyrgyzstan, Uzbekistan, Kazakhstan.A
Product Integrity and Approvals
Fluorouracil Injection is produced in accordance with WHO-GMP and ISO certifications, complying with United States Pharmacopeia guidelines. The solution is manufactured under stringent quality control standards, ensuring its efficacy and safety for cancer treatment applications. These certifications support the product's acceptance for dealer, distributor, exporter, manufacturer, supplier, trader, and wholesaler channels.
Clinical Use and Compatibility
This chemotherapy agent is indicated for a variety of cancers including colon, rectal, breast, stomach, and pancreatic malignancies. Its clear, colorless to faint yellow solution is compatible with commonly used intravenous fluids like 5% dextrose and 0.9% sodium chloride. Acidic solutions must be avoided, as they can affect product stability.
Administration and Storage Guidelines
Fluorouracil Injection is administered intravenously under strict medical supervision. Available in glass ampoules and vials of 10 mL and 20 mL, the product should be stored in a cool, dry place to maintain its 2-year shelf life. Proper handling and storage protect its pharmacological integrity and therapeutic effectiveness.
FAQ's of Fluorouracil Injection:
Q: How should Fluorouracil Injection be administered?
A: Fluorouracil Injection is administered intravenously by a healthcare professional, typically in a clinical setting such as a hospital or cancer treatment center. It should not be self-administered or given outside a medical facility due to potential risks and the need for patient monitoring.Q: What types of cancers is Fluorouracil Injection used to treat?
A: This medication is used as part of antineoplastic chemotherapy regimens, primarily for cancers such as colon, rectal, breast, stomach, and pancreatic cancer. Your oncologist will determine if this treatment is suitable for your type of cancer.Q: When should Fluorouracil Injection not be mixed or administered with other solutions?
A: Fluorouracil Injection should not be mixed with acidic solutions, as this can compromise the stability and efficacy of the drug. It is compatible with 5% dextrose and 0.9% sodium chloride solutions for intravenous administration.Q: Where should the product be stored to ensure maximum shelf life?
A: Store Fluorouracil Injection in a cool and dry place, away from direct sunlight and heat sources. Proper storage conditions help preserve the solution's potency and ensure it remains effective for up to 2 years.Q: What are the benefits of using Fluorouracil Injection in chemotherapy?
A: Fluorouracil Injection inhibits the growth of cancer cells, aiding in shrinking tumors and controlling disease progression. As a widely recognized and clinically validated antineoplastic agent, it forms an essential component of many chemotherapy protocols.Q: What precautions should be observed when using this injection?
A: Fluorouracil should only be used under the supervision of qualified healthcare personnel. Regular monitoring, dose adjustments, and supportive care are essential to minimize side effects and ensure safety during chemotherapy treatment.Q: What should I do if I miss a scheduled dose?
A: If you miss a scheduled dose, contact your healthcare provider immediately. Do not attempt to self-administer or reschedule without professional advice, as treatment timing and dose adjustments are critical for optimal outcomes.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
Other Products in 'Anticancer Injection' category
 |
SAINTROY LIFESCIENCE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |