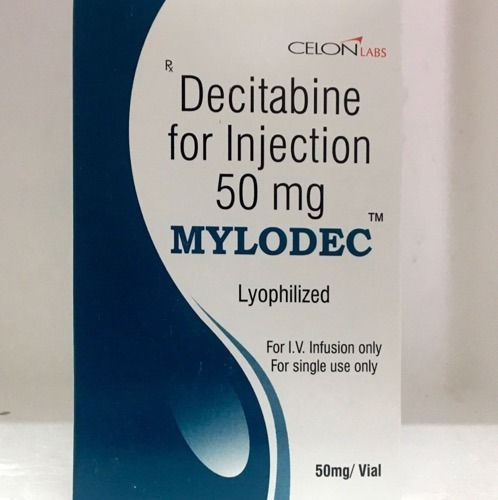
Decitabine For Injection
Product Details:
- Salt Composition Decitabine 50 mg per vial
- Indication Treatment of Myelodysplastic Syndromes (MDS)
- Dosage Form Lyophilized Powder for Injection
- Enzyme Types Other
- Feature Other
- Ingredients Other
- Application Other
- Click to View more
Decitabine For Injection Price And Quantity
- 100 Box
- 1 USD ($)/Box
Decitabine For Injection Product Specifications
- Other
- Decitabine 50 mg per vial
- Treatment of Myelodysplastic Syndromes (MDS)
- Other
- Other
- 4.0 - 7.0 (after reconstitution)
- Other
- Lyophilized Powder for Injection
- Normal Smell
- 2 Years
- White to off-white lyophilized powder
- Store at cool and dry place.
- Available under various licensed brands
- Pregnancy, known hypersensitivity to Decitabine or excipients
- Reconstitute with appropriate diluent and administer intravenously
- Reconstitute with 10 mL sterile water for injection. Further dilute in infusion fluid as per guidelines.
- Prescription only
- USP Type I glass vial
- Single-use glass vial with flip-off seal
Decitabine For Injection Trade Information
- surat
- Delivery Point (DP), Cash on Delivery (COD), Letter of Credit (L/C), Telegraphic Transfer (T/T), Western Union, Paypal, Letter of Credit at Sight (Sight L/C), Cash Against Delivery (CAD), Days after Acceptance (DA), Cash in Advance (CID), Cheque, Cash Advance (CA)
- 1000000 Box Per Month
- 4 Days
- Contact us for information regarding our sample policy
- 1 Vial
- Australia, North America, South America, Eastern Europe, Western Europe, Middle East, Asia, Central America, Africa
- Dadra and Nagar Haveli, Chandigarh, Himachal Pradesh, Andaman and Nicobar Islands, Uttarakhand, Daman and Diu, Lakshadweep, Nagaland, South India, East India, West India, Andhra Pradesh, Assam, Arunachal Pradesh, Bihar, Delhi, Gujarat, Goa, Haryana, Jammu and Kashmir, Jharkhand, Karnataka, Madhya Pradesh, Maharashtra, Mizoram, Meghalaya, Manipur, Pondicherry, Rajasthan, Sikkim, Tamil Nadu, Telangana, Tripura, Uttar Pradesh, West Bengal, North India, Punjab, Kerala, Central India, Odisha, Chhattisgarh, All India
- ISO/GMP/c-GMP/WHOGMP/USFDA/EUGMP.
Product Description
Since last manyyears Saintroy Lifescience is leading manufacture, export and supplierofDecitabine for Injectionfrom Surat, Gujarat, India. Decitabine forInjection used to treat myelodysplastic syndromes (MDS) including previously treated and untreated, de novo and secondary MDS of all French-American-British Subtypes.
Tags: Decitabine for Injection manufacturer, Decitabine forInjection exporter, Decitabine for Injection supplier, and Decitabine forInjection, whogmp approved Decitabine for Injection manufacturer and supplier, Decitabine ForInjection distributor, Decitabine for Injection shipper, Decitabine forInjection 50 mg Decitabine for Injection 50 mgsupplier,Decitabine for Injection 50 mg.
ISO/GMP/cGMP/WHOGMP/NAFDAC approved, PPB Kenya approved/USFDA approved/EUGMP approved/MOH Iran approved manufacturingfacility inGujarat, India.
Registration: Saintroy Lifescience is interested inregistration of own brand or customer brands in ministry of health of clientscountry and we also provide the products samples, COA, COPP, ACTorCTD dossier, DMF, Manufacturing license, Free sales certificate and other required documents.
Export Country :Saintroy Lifescience Decitabine forInjection export to USA, Canada, Germany, Europe, France, Ghana, Nigeria,Kenya, Senegal, Sudan,UAE, Kuwait, Dubai, Cameroon, Lebanon, Peru, Chile,Somalia, Philippines, Myanmar, Maldives, Thailand, Singapore, Malaysia, HongKong, China, Zimbabwe, Togo, Burkina Faso, Niger,Guinea Bissau, Congo,Democratic of republic, Ivory Costa, Oman, Guatemala, Nicaragua, Salvador,Qatar, Costa Rica, Panama, Cuba, Colombia, Bolivia, Argentine, Algeria, Guyana,Morocco, Jordan, Bahrain, Iran, Egypt, Kyrgyzstan, Uzbekistan, Kazakhstan.
Comprehensive Cancer Therapy Solution
Decitabine For Injection is formulated as a lyophilized powder, providing an effective intravenous treatment for patients diagnosed with Myelodysplastic Syndromes (MDS). With a shelf life of 2 years and a normal fermentation smell, this medication offers reliability and stability for clinical settings, including hospitals and infusion centers.
User-Friendly Packaging for Medical Professionals
Each dose comes in a single-use USP Type I glass vial with a secure flip-off seal, designed to uphold pharmaceutical standards and prevent contamination. The vial's packaging ensures ease of handling and accurate dose preparation, making it suitable for dealer, distributor, exporter, manufacturer, supplier, trader, and wholesaler networks across India.
FAQ's of Decitabine For Injection:
Q: How should Decitabine For Injection be prepared and administered?
A: Reconstitute the lyophilized powder by adding 10 mL of sterile water for injection to the vial. Gently swirl to dissolve, then further dilute the solution in the appropriate infusion fluid as per clinical guidelines. The medication should only be administered intravenously by a healthcare professional.Q: What are the primary indications for using Decitabine For Injection?
A: This medication is indicated for the treatment of Myelodysplastic Syndromes (MDS) and must be prescribed by a qualified physician. It is not intended for self-administration or for conditions outside its approved use.Q: When should Decitabine For Injection not be used?
A: Decitabine For Injection is contraindicated in pregnant individuals, and in patients with known hypersensitivity to Decitabine or any of its excipients. Always consult your physician regarding suitability before starting treatment.Q: Where should Decitabine For Injection be stored?
A: The vial should be kept in a cool and dry place to maintain its stability and efficacy. Always store as directed in the product guidelines and keep out of reach of children.Q: What is the physical appearance and composition of Decitabine For Injection?
A: It is supplied as a white to off-white lyophilized powder in a single-use vial. Each vial contains 50 mg of Decitabine and other ingredients, with a pH level of 4.0-7.0 after reconstitution, and possesses a normal fermentative odor.Q: How does the packaging ensure product safety and quality?
A: The USP Type I glass vial with a flip-off seal offers superior protection against contamination and pharmaceutical degradation, supporting both product safety and ease of use for medical professionals.Q: What benefits does Decitabine For Injection offer to healthcare providers and patients?
A: This product delivers a standardized, clinically validated treatment for MDS, backed by a reliable supply chain for dealers, distributors, exporters, and healthcare institutions in India. Its ready-to-use format and secure packaging streamline preparation and administration.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
Other Products in 'Anticancer Injection' category
 |
SAINTROY LIFESCIENCE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |