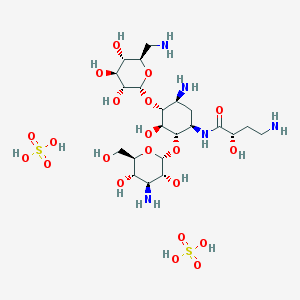
Amikacin Sulphate
1-5 USD ($)
Product Details:
- Taste Bitter
- Ph Level 4.5 - 6.5 (1% solution)
- Molecular Formula C22H47N5O21S2
- HS Code 29419090
- Loss on Drying Not more than 4.5%
- Structural Formula (Refer to chemical literature or product documentation)
- Color White to off-white
- Click to View more
X
Amikacin Sulphate Price And Quantity
- 5 Kilograms
- 1-5 USD ($)
Amikacin Sulphate Product Specifications
- C22H47N5O21S2
- 4.5 - 6.5 (1% solution)
- Used to treat severe bacterial infections
- 29419090
- Not more than 4.5%
- White to off-white powder
- AMIKIN
- >99%
- Bitter
- Odorless
- As per specification / customizable
- >200C (decomposes)
- Solid
- AMIKACIN SULPHATE
- Not more than 0.001%
- 781.752 g/mol Grams (g)
- 254-711-8
- Non-poisonous under normal conditions
- 39831-55-5
- Pharmaceutical Grade
- Store in a cool, dry place, protected from light
- Freely soluble in water, practically insoluble in alcohol and chloroform
- Antibiotic API
- (Refer to chemical literature or product documentation)
- White to off-white
- 3 years from date of manufacture
- <0.5 IU/mg
- +94 to +100 (C=1, Water)
- Not more than 1.0%
- Not less than 900 g/mg of C22H43N5O13S
- Not more than 12.0% (by KF Titration)
- 4.5 - 6.5
- Complies as per pharmacopeia
- <0.5 IU/mg
- Passes
- Complies with pharmacopeial standards
Amikacin Sulphate Trade Information
- Nhava sheva Port, Hajira Port, Mundra Port.
- Paypal, Letter of Credit (L/C), Western Union, Letter of Credit at Sight (Sight L/C), Telegraphic Transfer (T/T), Cash in Advance (CID), Cash Advance (CA)
- 10000 Kilograms Per Month
- 6 Days
- Yes
- If order is confirmed we will reimburse the sample cost
- 25 KG DRUM PACKING
- All India
- EUGMP/WHOGMP/GMP.
Product Description
Amikacin Sulfate is the sulfate salt of amikacin, a broad-spectrum semi-synthetic aminoglycoside antibiotic, derived from kanamycin with antimicrobial property. Amikacin irreversibly binds to the bacterial 30S ribosomal subunit, specifically in contact with 16S rRNA and S12 protein within the 30S subunit. This leads to interference with translational initiation complex and misreading of mRNA, thereby hampering protein synthesis and resulting in bactericidal effect. This agent is usually used in short-term treatment of serious infections due to susceptible strains of Gram-negative bacteria.
Tags :- Amikacin Sulphate Manufacturer, Amikacin Sulphate Manufacturer in India, Amikacin Sulphate Supplier, Supplier in India, AmikacinSulphate Distributor, Amikacin Sulphate Distributor in India, Amikacin SulphateMerchant Exporter, Amikacin Sulphate Exporter, Amikacin Sulphate Exporter inIndia, api, apis, Pharmaceutical Ingredients, Active PharmaceuticalIngredients, Amikacin Sulphate Bulk Drugs manufacturer, Amikacin Sulphate BulkDrugs, Amikacin Sulphate Bulk drugs in India, Amikacin Sulphate Api in India,Bulk Drugs, Bulk Drugs in India.
Comprehensive Quality Standards
Amikacin Sulphate easily passes pharmaceutical-grade standards, evidenced by its excellent assay results and compliance with endotoxin, pH, heavy metal, and residue controls. Each batch undergoes thorough testing, ensuring purity exceeds 99% and water content stays under 12%. These controls guarantee consistent performance for medical use.
Primary Uses and Benefits
This antibiotic is critical in the management of severe bacterial infections, particularly when other treatments are ineffective. Its potent antibacterial action and favorable solubility in water make it a reliable choice for tailored pharmaceutical formulations. Healthcare providers rely on Amikacin Sulphate both for effectiveness and safety in serious clinical settings.
Handling, Storage, and Shelf Life
Store Amikacin Sulphate in a cool, dry place away from direct light to preserve its stability and potency. Under proper storage, the product maintains its efficacy for up to three years from the date of manufacture. Its stable, non-poisonous nature makes it manageable in most pharmaceutical environments.
FAQ's of Amikacin Sulphate:
Q: How is Amikacin Sulphate typically used in medical treatments?
A: Amikacin Sulphate serves as an active pharmaceutical ingredient in injectable antibiotics designed for severe bacterial infections. It is administered under medical supervision as part of hospital-based therapies, especially when other antibiotics fail.Q: What quality standards does Amikacin Sulphate comply with?
A: Each batch is rigorously tested to comply with pharmacopeial standards, including limits on residue on ignition (<1.0%), endotoxin levels (<0.5 IU/mg), pH (4.5-6.5), water content (<12.0%), and heavy metals (<0.001%). Related substances and identification tests are also carried out to ensure purity.Q: When should Amikacin Sulphate be used over other antibiotics?
A: This compound is preferred when bacterial infections are severe or resistant to other antibiotics. It is used according to doctor's recommendation, particularly in hospital settings where potent, broad-spectrum activity is necessary.Q: Where should Amikacin Sulphate be stored to maintain its stability?
A: Store the powder in a cool, dry place, and keep it protected from light. Proper storage is essential to maintain its efficacy and maximize its three-year shelf life.Q: What is the process for verifying the purity and quality of Amikacin Sulphate?
A: Purity and quality are assessed through HPLC assay (not less than 900 g/mg of C22H43N5O13S), microbiological testing, endotoxin level checks, specific optical rotation, and various compliance tests as stipulated by pharmacopeial standards.Q: What benefits does Amikacin Sulphate offer to manufacturers and healthcare providers?
A: The product assures consistent pharmacological performance, high purity, and reliable compliance with international quality standards, making it a preferred choice for safe and effective pharmaceutical production and patient care.Q: Is Amikacin Sulphate toxic or harmful to handle in standard pharmaceutical environments?
A: It is considered non-poisonous under normal conditions and can be safely handled and processed in well-regulated pharmaceutical settings when standard hygiene and protective measures are followed.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'Active Pharmaceutical Ingredients' category
 |
SAINTROY LIFESCIENCE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |